Electron affinity definition


It is either applied to an atom or a molecule. Given below is the table that consists of the detailed differences of both the terms.ĭifference Between Electronegativity and Electron AffinityĮlectronegativity refers to the ability of the atoms to attract the electrons from the other elements.Įlectron affinity refers to the amount of energy that is liberated whenever a molecule or a neutral atom tends to acquire an electron from the other elements.
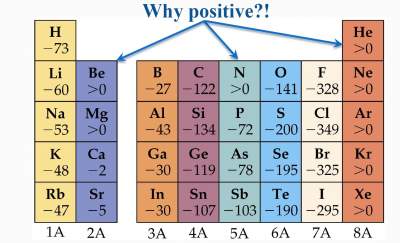
Understanding the differences between electronegativity and electron affinity is important so that we do not use them interchangeably. In this article, we will study about electronegativity vs electron affinity in detail. However, electronegativity is the result of the property of attraction, while electron affinity is the result of the change in energy. Both electronegativity and electron affinity deal with the movement of the electrons. These two properties are often known to be interrelated but they are not interchangeable. On the other hand, electron affinity is related to the release of the energy whenever an electron tends to get added to an atom. The primary difference between electronegativity and electron affinity lies in the fact that electronegativity is the property related to the ability of an electron to attract towards an atom. Both electronegativity and electron affinity are two different chemical properties that are associated with the elements.
